- Request access to the controlled data sets from the appropriate dbGaP (Database of Genotypes and Phenotypes) Data Access Committee (DAC): https://dbgap.ncbi.nlm.nih.gov/aa/dbgap_request_process.pdf.
- Download and complete the model Data Use Certification (DUC) for the controlled data set of interest:
- Visit the dbGAP site at http://www.ncbi.nlm.nih.gov/gap.
- Search for the study from which data are requested. For example, “joint addiction, aging, mental health” results in number of studies.
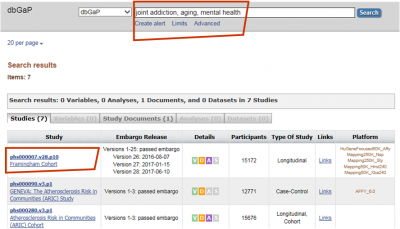
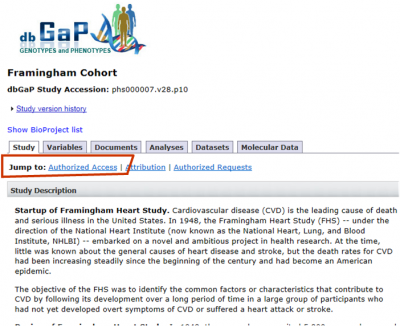
- Click on the linked study title of interest for instructions on how to download the model DUC and determine if IRB approval is required. Model DUC and IRB Requirements can be found under the “Study” tab in the “Authorized Access” section (as shown in screenshot).
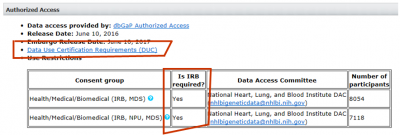
- If IRB approval is required, go to the section for “dbGaP Access Request” found at https://ovpr.uchc.edu//rcs/hspp/irb/irb-instructions-forms-and-samples/
download and complete the form “dbGaP Access Request Form for IRB Certification.”
- Complete the Data Security and Data Release Form (MS Word) (PDF).
- Email the completed Model Data Use Certification, IRB Approval for dbGaP Data Access (if required), and Data Security and Data Release Reporting form to the AVP for Research, Research IT Services – Dr. Khamis Abu-Hasaballah – at khamis@uconn.edu. Upon verification and approval of these documents, the AVP for Research will issue a certification letter signed by him, the Institutional Chief Security Officer, and the Institutional Signing Official (SPS Director).
uconn health
Health Center Research Advisory Council (HCRAC) Contacts
Health Center Research Advisory Council
UConn Health
263 Farmington Avenue
Farmington, CT 06030-6403
For questions or comments about the HCRAC please contact one of the following individuals.
| Name | Title | Phone | |
|---|---|---|---|
| Christopher Heinen | Council Chair | (860) 679-8859 | cheinen@uchc.edu |
| Stephanie Holden | HCRAC Coordinator | (860) 679-2513 | sholden@uchc.edu |
Travel Grants for Postdoctoral Fellows
Application Guidelines and Instructions
This program has been established to support travel costs for postdoctoral fellows who present research results in-person at scientific meetings and symposia. The program will support costs of that travel up to a limit of $700 per fiscal year. Note, HCRAC travel funds cannot be combined with any other UConn travel support.
Awards are made on a first-come, first-served basis, until the funds for this program have been exhausted.
Eligibility
- Eligibility for this program is restricted to UConn Health postdoctoral fellows whose faculty preceptor for research is based at UConn Health and contributes their indirect costs to UConn Health.
- Applicants must be first author on a research abstract and present a corresponding poster or talk; the topic must be central to postdoctoral research performed at UConn Health.
- Postdocs may only receive one travel grant in a given year .
If there are any questions about this travel grant program, please contact Stephanie Holden at 860.679.2513 or sholden@uchc.edu.
InfoEd eRA Portal
 The UConn Health InfoEd is an enterprise-level, web-based application designed to manage all activities related to the management and execution of the research project life cycle – from cradle to grave. The system comprises a suite of modules, each designed to manage a specific aspect of the research project cycle. These include: proposal development and submission to the funding entity during the pre-award phase (Proposal Development – PD) ; proposal tracking and management in the post-award phase (Proposal Tracking – PT); human research protocol application management (Human Subjects Management – HSM); lab animal protocol application management (Lab Animals Management – LAM); environmental safety management (Environmental Safety Management – ESM); intellectual property management (Technology Transfer – TT); and clinical trial management, case report form (CRF) design, and scientific data collection and management (Clinical Trials Management – CTM). Modules currently installed at UConn Health include:
The UConn Health InfoEd is an enterprise-level, web-based application designed to manage all activities related to the management and execution of the research project life cycle – from cradle to grave. The system comprises a suite of modules, each designed to manage a specific aspect of the research project cycle. These include: proposal development and submission to the funding entity during the pre-award phase (Proposal Development – PD) ; proposal tracking and management in the post-award phase (Proposal Tracking – PT); human research protocol application management (Human Subjects Management – HSM); lab animal protocol application management (Lab Animals Management – LAM); environmental safety management (Environmental Safety Management – ESM); intellectual property management (Technology Transfer – TT); and clinical trial management, case report form (CRF) design, and scientific data collection and management (Clinical Trials Management – CTM). Modules currently installed at UConn Health include:
- Proposal Tracking (PT) – This module acts as a central clearinghouse of both pre- and post-award information. It provides a single reference point for tracking all details related to proposals including: budgets, subcontracts, approvals, technical reports, and all associated communications.
- Financial Conflict of Interests – This module provides researchers with the ability to complete and submit researcher and staff financial interest disclosures electronically. It allows Research Compliance Services staff to track and manage conflicts of interest, establish management plans, and fulfill our policy requirements. The module interfaces with proposals and human subjects. These materials include how-to instructions for UConn Health. Consult the FCOI website at UConn Storrs for the UConn Storrs and Regional Campuses FCOI how-to materials.
- Animal Facility Management / CCM Module – The InfoEd Animal Facility Management supports the animal care operations of the Center for Comparative Medicine (CCM) including animal ordering, husbandry, census, and billing of animal facility protocols.
- Lab Animal Use / IACUC Module – This module is designed to support the review and management of animal research protocols by the Institutional Animal Care and Use Committee (IACUC).
- Tech Transfer (TT) – This module was used to manage administrative data relating to intellectual property and tracks inventors, patents, key dates, contacts, agreements, documents and communications, and financial information tied to expenses and royalties. Now it contains legacy data on some University IP and was replaced by Wellspring Sophia.
- SPIN (Sponsored Programs Information Network) – SPIN is a searchable database that provides real-time access to current research funding opportunities. All data in SPIN is obtained directly from the sponsoring agencies themselves to ensure authenticity. To use SPIN, login to InfoEd and then choose Find Funding link at the top of the page.
- SMARTS (SPIN Matching And Research Transmittal Service) – SMARTS is an automated system that periodically queries the SPIN database and sends notifications to investigators regarding available funding opportunities in their field of study.
Sponsored Program Administration (SPA) Archive
- March 18, 2026 – Presentation Slides
- Agenda
- Export Control & Research Security – Mike Shelton
- Pre-Award
- Updated Elements of an NIH Data Management and Sharing Plan
- Fellowship Budgets
- Federal Sponsor Updates
- Proposed Deadlines
- Post-Award
- Pre-Award Requests
- Banner Upgrades
- Training
- New job aid
- Federal Costing Principles training
- Training Resources
- February 18, 2026 – Presentation Slides
- Agenda
- Policy Update: Policy on Alleged Misconduct in Research – Meg Johnson
- Pre-Award
- Federal Sponsor Updates
- Foreign Subaward
- Direct Costs Greater than $500K
- Salary Cap
- Common Forms
- Malign Foreign Talent Recruitment Program
- Proposal Timeline Policy
- Federal Sponsor Updates
- Post-Award
- DHHS Update – Salary Cap
- Award Closeout Transfer Voucher Process
- Stipends on Sponsored Awards
- Training
- The Abstract – newsletter
- New LevelUp Module – AOR Fundamentals: From Proposal Submission to Closeout
- SPA Training Resources
- Agenda
- January 21, 2026 – Presentation Slides
- Agenda
- Pre-Award
- NIH: Biographical Sketch & Current and Pending Other Support Forms
- National Science Foundation: Policy Update
- Post-Award
- Guidelines for Faculty Maximum Effort on Sponsored Projects
- Training
- New LevelUp Modules
- NYC Research Administration Demonstration Series
- SPA Training Resources
- Pre-Award
- Agenda
- November 19, 2025 – Presentation Slides
- Agenda
- Research Security and Export Control – Lesley Salafia
- Pre-Award
- Proposal Submission
- Interim Guidance on Reopening of NIH Extramural Activities
- Post-Award
- Subawards Submission Tips and Tricks
- Student Fees
- Training
- LevelUp
- SPA Training Resources
- Agenda
- October 22, 2025 – Presentation Slides
- Agenda
- Financial Conflicts of Interest in Research – Meg Johnson
- Pre-Award
- Research Security training
- NIH: Other Support / Research Security Training
- Information and Compliance Form for Subrecipients
- Post-Award
- Outstanding Checks
- Travel Allocation
- SPA Training
- LevelUp updates
- SPA Training – New Resources
- Agenda
- September 17, 2025 – Presentation Slides
- Agenda
- IRB Updates – Shemetra Owens
- Pre-Award
- Research Security training
- NIH limiting submissions to 6 per calendar year effective 9/25/25
- Updated NIH NCE policy
- Post-Award
- Banner Budget – Travel
- Shipping / Express mail fees on awards
- Effort Reporting Audit – Early Outcomes
- SPA Training
- LevelUp updates
- New e-Learning modules now available in Saba
- SPA Training resources
- Training Resources job aid / website tour
- Agenda
- June 18, 2025 – Presentation Slides
- Agenda
- Introducing – Meg Johnson, Director of Conflicts of Interest and Research Integrity
- Research Finance
- Capital purchases – Capital Procurement Account Guidance
- Transfer vouchers
- Pre-Award
- Internal Forms: IPR and Subrecipient Forms Updated
- NIH updates
- NOFO Solicitation Requirements – Reminder
- PA-23-272—Parent F31 – Individual Predoctoral Fellowship update
- Post-Award
- Outstanding Checks
- AMS Rebudget process
- SPA Training
- 2025-2026 SPA Meetings – opinion poll
- Upcoming NCURA webinar – Research Administration in a Changing Federal Landscape
- Budgeting Basics e-learning course
- SPA Training resources
- Agenda
- May 21, 2025 – Presentation Slides
- Agenda
- Pre-Award
- Biographical Sketch: Adoption of the Common Form
- NSF: Implementation of Standard 15% IDC
- Foreign Subawards
- NIH No-Cost Extensions
- Research Security Training
- SPS Forms – Changes coming
- Post-Award
- Staffing Updates
- FY 2025 NRSA Stipend / Tuition
- Year End Reminders
- Executive Order – 14222
- Training
- LevelUP update
- New jobs aids – Budgeting Basics and Effort Reporting
- Training resources
- Pre-Award
- Agenda
- April 16, 2025 – Presentation Slides
- Agenda
- Pre-Award
- DHHS/NIH Update – Salary Cap
- Docusign – 5-Day Submission Policy
- 50K Program
- NIH Common Forms – Postponed
- Post-Award
- 2024 Single Audit
- Year End Reminders
- SPA Training Team
- LevelUp Update
- New EPAF update
- Training Resources
- Pre-Award
- Agenda
- March 19, 2025 – Presentation Slides
- Agenda
- Research Security Training Deadline – Lesley Salafia
- Pre-Award
- Federal Sponsor Updates
- NIH Common Forms (Required in May)
- NIH Fellowships – Forms-H
- Federal Sponsor Updates
- Post-Award
- DHHS Salary Cap Update
- Transfer Voucher Guide
- SPA Training Team
- Policy Manager Demo
- LevelUp Updates
- Training Resources
- Agenda
- January 15, 2025 – Presentation Slides
- Agenda
- Pre-Award
- NIH: FORMS-I
- Changes to NIH and AHRQ Fellowship Applications
- Pre-Award
- Agenda
-
-
- Post-Award
- FY 2025 Q2 Effort Reports
- Accounting for Costs – Uniform Guidance
- Accounting for Costs – Cost Accounting Standards (CAS)
- Accounting for Costs – HuskyBuy/Banner
- Training
- LevelUp updates
- New NoA Job aid
- Training resources
- Post-Award
-
- November 18, 2024 – Presentation Slides
- Agenda
- Financial Disclosures in Research – Gus Fernandez-Wolff
- Pre-Award
- Post-Award
- Food Related Expenses on Sponsored Awards
- Payroll & LDCA
- Staffing Update
- SPA Training
- SRA Modules & Training Initiative
- NIH Upcoming Events
- NSF Fall 2024 Virtual Grants Conference
- Agenda
- October 16, 2024 – Presentation Slides
- Agenda
- UCH Biosafety Program – Victoria Scranton, Biosafety Program Coordinator
- Pre-Award
- NIH – New Data Management and Sharing (DMS) Policy Questions in RPPR
- New DMS Section in Just-in-Time
- New ‘DMS Request’ in Prior Approval
- NIH Resources
- Acceptable/Unacceptable Signature Formats – Other Support pages
- Post-Award
- LDCA – By the Numbers
- AMS Rebudget
- Training
- Upcoming NIH Events
- New NCURA videos available on the website: The RPPR Matrix and Culture of Compliance
- Agenda
- September 18, 2024 – Presentation Slides
- June 26, 2024 – Presentation Slides
- Research Security & Undue Foreign Influence – NSPM-33 and CHIPS Act Requirements
-
- Agenda
- Export Control Overview – Mike Shelton
- Banner 9 upgrade
- Post-award
- FY 2024 NRSA Stipend / Tuition levels
- Year End Reminders
- Quick hits – Travel Authorizations
- Malign Foreign Talent Recruitment Program
- SPA Training – June meeting
- Agenda
-
- Agenda
- Foreign Travel / Export Control Updates – Mike Shelton and Bieu Tran
- Pre-award
- Federal Sponsor Updates - NSF
- Post-award
- 2023 Single Audit
- FY 2024 NRSA Stipends – NOT-OD-24-059
- Banner Upgrade
- P-Card Review
- Agenda
- March 20, 2024 – Presentation Slides
- Agenda
- Open Mike
- Financial Disclosure in Research- Gus Fernandez-Wolff
- Research Security – Mike Rock
- Pre-award
- Federal Sponsor Updates
- IPR Routing Form
- IPR Routing Form – PI Certification
- Agenda
- February 21, 2024 – Presentation Slides
- Agenda
- SPA Training announcements
- Pre-award
- Understanding NIH Application/Grant Numbers
- NIH Guidance on Marking Changes in Resubmission Applications
- NIH/DHHS Salary Limitation 2024
- Delegations in eRA Commons and RPPR Privileges
- Reports Due to Sponsor
- Post-award
- EPAFs, Tips, Tricks & Reminders
- Award Budget Reminder – Travel
- Agenda
- January 17, 2024 – Presentation Slides
- Agenda
- SPA Training announcements
- New On-Demand e-learning modules
- SPA Training Resources
- Pre-award
- Federal Sponsor Updates – NIH
- NIH Letters of Support
- OVPR Proposal Submission Policy
- Fellowship Applications – Submission Policy
- Post-award
- Cost Accounting Standards – 9905.502 – Banner Accounts
- AMS Rebudget Module – slides
- SPA Training announcements
- Agenda
- November 15, 2023 – Presentation Slides
- Agenda
- InfoEd version 13 (v13) Portal View
- Research Finance – F & A Base Year Follow-Up
- Tracking Students & Unpaid Occupants
- Preliminary Inventory Review
- Pre-award
- NIH Foreign Subrecipients – access to documents requirement
- Post-award
- Cost Transfer Best Practices
- SPA Training
- NCURA Region 1 – Spring meeting
- Resources
- December SPA
- Agenda
- October 18, 2023 – Presentation Slides
- Agenda
- Introducing the DMPTool – Adria Barbano, Academic IT Services
- Research Finance
- Reallocating Grant Fringe Budgets
- F & A Proposal- Base Year 2024
- Pre-award
- Changes to DSMP – Prior Approval
- Sample Routing Documents
- Proposal and Award Budget Templates
- Proposal Submission Policy
- New administrative staff – email lists
- Inform Your Faculty – reminder
- Cost Share Approval
- IBC and ICUC Grant Congruence Review Process Change
- Post-award
- F & A on Sponsored Projects
- SPA Training resources
- Agenda
- September 20, 2023 – Presentation Slides
- Agenda
- Pre-award
- DMPTool
- NIH RPPR – Section D
- NIH Data Management and Sharing Costs
- NIH – Hyperlinks Unallowable
- PI Eligibility – Fellowship Applications
- Proposal and Award Budget Templates
- Inform Your Faculty
- Research Fringe Benefits rates
- AMS Updates
- Post-award
- Reminders and New Processes for Better Documenting Cost Allocation
- Consulting Invoice Checklist
- Procurement/Contracts Information Session – 10/30, 1:00 p.m., Keller Auditorium
- SPA Training – Upcoming classes
- Pre-award
- Agenda
- May 17, 2023 – Presentation Slides
- Agenda
- Introduction – Bernard Cook, School of Medicine
- Updates to Funding Opportunity Terminology
- Policy – Qualifying Submissions
- Change to NIAMS Participation in NIH Parent R21 Announcements
- Year-End Reminders
- Training – class attendance
- Upcoming training classes
- Agenda
- April 19, 2023 – Presentation Slides
- Agenda
- eRA Commons News – Redesigned xTRAIN
- Compliance Reminder
- NIH Other Support Reminders
- IPR Compliance Crosswalk
- SPS Staffing Update
- 75521 Computing Devices – Guidance
- AMS Events
- SPA training – upcoming classes
- Agenda
- March 15, 2023 – Presentation Slides
- Agenda
- NIH Data Management and Sharing Policy – Reminders
- Proposal Tips and Tricks
- Indirect Costs on Cost-Shared Expenses
- Participant Support vs. Human Subjects Payments
- SPS Staffing Update
- Pre-Award Requests
- LDCA’s
- Account Code 75520
- SPA Training – upcoming classes
- Agenda
- February 22, 2023 – Presentation Slides
- Agenda
- Proposal Submission Policy
- Assist Applications
- eRA Commons Accounts – Personal Profile
- UCH as a subrecipient – reminders
- NSF Safe & Inclusive Workspaces – New Requirement
- NSF Audit Reminders
- FY 2023 NRSA Stipend/Tuition Levels
- Student Fees
- No Cost Extensions – Best Practices
- New Policies and where to find them
- CRA study group
- SPA Training Job Aids
- Agenda
- January 25, 2023 – Presentation Slides
- Agenda
- Confidentiality Agreements and Non-Disclosure Agreements
- SPS Staff Updates
- NIH Data Sharing and Management Plan Requirement
- NIH/DHHS Salary Limitation 2023
- NIH Project Roles
- Eligibility to Serve as PI
- Proposal Tips and Tricks – Cash vs. In-Kind Cost Share
- SPA Training Updates
- Agenda
- November 16, 2022 – Presentation Slides
- Agenda
- InfoEd updates
- OVPR Proposal Submission Policy
- Sample Routing Documents
- Proposal Routing Requirements Checklist
- ABP/Other Bonuses
- A/P Invoicing
- Subaward Requests in HuskyBuy
- AMS Events Module
- SPS Updates
- Overview of Agreements reviewed/negotiated by Contracts Group
- SPA Training Announcements and Updates
- Agenda
- October 19, 2022 – Presentation Slides
- Agenda
- IRB Presentation – Mayra Cagganello
- NASA Certification
- NIH Data Sharing & Management Plan
- Contract Submission Reminders
- Annual Data Report
- SPA Training – upcoming classes
- Agenda
- September 21, 2022 – Presentation Slides
- Agenda
- Environmental Health and Safety Updates
- Grants.gov Downtime
- NIH Genomic Data Sharing
- NCI Minimum Level of Effort
- NSF Fastlane
- Mobile Device Management
- Contract Services Group – updates
- SPA Training Updates
- Agenda
- June 22, 2022 – Presentation Slides
- Agenda
- Accounting for Costs – UG, CAS, HuskyBuy/Banner
- DHHS F & A Rate Agreement
- eRA Commons Enhancements
- Proposal Budget Template
- SPS Personnel Updates
- SPS Contact Assignments
- Greenphire update
- Agenda
- May 18, 2022 – Presentation Slides
- Agenda
- SPA Announcement
- June 2022 Updated RPPR Module and Instructions
- Internal Routing and Submission Deadlines
- Contracted Salary Increases – Budgets for Proposals
- Institutional Base Salary
- FY 2022 NRSA Stipend/Tuition Levels
- Agenda
- April 20, 2022 – Presentation Slides
- Agenda
- HuskyBuy Invoice Approval Automation
- Year end calendar
- New onboarding process for unpaid experiential
- AMS go-live update
- Personnel changes
- New fringe benefits rates to use for budgeting
- Proposal Routing – Documents and Timeline
- Flatten PDFs – JIT and RPPR
- Agenda
- March 16, 2022 – Presentation Slides
- Agenda
- 50K Program
- SPA Training updates – website updates and new Level One Basics class
- DocuSign updates
- DocuSign – resource discussion
- Other Support/In Kind Support FAQ’s
- Agenda
- February 16, 2022 – Presentation Slides
- Agenda
- PI Eligibility Fellowship applications
- New IPR Form
- DHHS/NIH Update – Salary Cap
- Subaward Invoice Review
- Mid Year Review
- Leadership update
- Agenda
- January 19, 2022 – Presentation Slides
- Agenda
- F&A Rate Agreement Renewal
- SPA Training
- NIH Era Commons Id For All Senior/Key Personnel
- NIH Submission Of Inclusion Enrollment Data
- NIH Annotated Forms G Set
- NIH Pre- And Post-Award Disclosures
- NIH Grants Policy Statement
- Uncashed Checks
- FDP Clearinghouse
- Agenda
- December 15, 2021 – Presentation Slides
- Agenda
- NIH New Requirements Updates
- NIH Prior Approval-Carryover-SPS Process
- Research Grant Reporting Request
- OVPR Routing Policy
- Agenda
- November 17, 2021 – Presentation Slides
- Agenda
- Training Update
- Open Mike
- Clinical Trials
- NIH Updates
- Agenda
- October 20, 2021 – Presentation Slides
- Agenda
- Research Development Services
- Banner 9 Upgrade
- Fall 2021 NIH Virtual Seminar on Program Funding and Grants Administration Registration
- eRA Commons Enhancement for Submit Reference Letter Screen
- Change to NIDCD Admin Reduction Guidance for Modular R01 Awards
- Proposal Dashboard Update
- Effort Certification After End Date
- Black Friday Update
- Agenda
- September 15, 2021 – Presentation Slides
- Agenda
- Fall 2021 NIH Virtual Seminar on Program Funding and Grants Administration Registration
- Unique Entity Identifier for Federal Sponsors
- NIH Updated Policy for Family Leave and Unpaid Leave for Extramural Loan Repayment Program
- NSF Website Enhancements
- Extending the Special Exception to the NIH/AHRQ/NIOSH Post Submission Material Policy During COVID-19
- NSF Pre-Award and Post Award Disclosures Related to the Bio-Sketch
- Scope of Work Change
- Approved Fringe Benefit Rates for 2022
- Agenda
- June 15, 2021 – Presentation Slides
- Agenda
- Institutional Base Pay
- NSF Publications Repository Changes
- New NIH Inbox for Biographical Sketch and Other Support Questions
- Revised NIH Grants Policy Statement
- eRA Commons Expanding Requirements for IDs
- What’s an Application Packet and When is it Needed
- Proposal Submission 9:00 am Due Date
- Agenda
- May 19, 2021 – Presentation Slides
- Agenda
- Biosafety Cabinet Certifications
- Year End Reminders
- Proposal Submission Policy Timeline and Reminders
- Fringe Benefit Rates
- NIH Biographical Sketch and Other Support
- Training Reminders
- Agenda
- April 21, 2021 – Presentation Slides
- Agenda
- Research Development Services Overview
- Salary Cap
- American Heart Association Carryover and No Cost Extension Updates and Reminders
- Publications and Printing Costs on Sponsored Awards
- IPR Routing Tips
- eRACommons Phasing Out Internet Explorer
- Redesigned RePORTTool
- eRACommons Login Tip
- NIH Biographical Sketch and Other Support
- Agenda
- March 17, 2021 – Presentation Slides
- Agenda
- NIH Update
- SPS Metrics YTD Comparison-Proposals/Awards
- Proposal Deadline Implementation
- Agenda
- February 17, 2021 – Presentation Slides
- Agenda
- IBC Discussion with Allison Pohl, Victoria Scranton, and Ron Wallace
- Student Fees
- PreAward Setup
- Updating PTR Before Hire
- NRSA
- Salary Cap
- Agenda
- December 16, 2020 – Presentation Slides
- Agenda
- Foreign Influence – Wesley Byerly
- OCTR Manager Change
- January Federal Costing Principles Module
- SPA Meeting Calendar Request
- ERA Commons Update
- Proposal Submission Policy
- Agenda
- November 18, 2020 – Presentation Slides
- Agenda
- MS SSRS Reports Update
- IPAS Discussion
- UConn Sponsored Program Metrics
- OVPR Proposal Submission Faculty Survey Results
- Proposal Submission Update
- Agenda
- October 21, 2020 – Presentation Slides
- Agenda
- Proposal Process – Under Development
- F&A on Projects of $50K or Less
- Sabbatical
- Principal Investigator Eligibility
- Use of Hypertext in NIH Grant Applications
- NIH Virtual Seminar
- OVPR Website
- Subaward Invoice Review
- Effort Reporting During Covid
- Agenda
- July 29, 2020 – Presentation Slides
- Agenda
- NIH updates
- NSF changes to award terms and conditions
- Spotlight on the new guidance for expenditures on sponsored awards
- NSF audit findings discussion
- Use of ASSIST
- gov resources
- Training videos
- Learn grants
- Visa costs
- Human subject participant payments
- Agenda
- June 24, 2020 – Presentation Slides
- Agenda
- OMB Expired Administrative Flexibilities
- HR Guidance Regarding Pay and Telecommuting
- Expenditures on Sponsored Awards Guidance
- Changes in HSS Post-Submission Updates with Forms-F
- PHS Human Subjects & Clinical Trials Information Form
- NIH, AHRQ, and NIOSH Updates
- Budgeting for Fringe Benefits on Sponsored Programs Updates
- OMB Expired Administrative Flexibilities
- Agenda
- May 26, 2020 – Presentation Slides
- Agenda
- Year-end Processing
- New Fringe Benefits Rates
- NIH Transition to Forms F
- COVID-related Information in RPPRs
- Requisitions needing SPS approval
- PPE Purchases
- Agenda
- April 16, 2020 – Presentation Slides
- Agenda
- COVID-19 Information
- Unique Entity Identifier
- NIH Updates
- NSF Updates
- FCOI Update
- Budget and Justification
- End of Grant
- Program Income
- Cost Transfers
- Subaward Invoice Review
- Fringe Rates
- Questions
- Agenda
- February 18, 2020 – Presentation Slides
- Agenda:
- Infographics Overview
- NIH Applications
- Change in Submission Deadlines and End of Recent Substantial Service Option
- Guidance on Video Submission
- SPS Routing Deadlines
- Certificate of Confidentiality
- Issuance of a Revised NSF Proposal and Award Policies and Procedures Guide
- DHHS Salary Cap
- NRSA Stipend/Tuition Levels
- Cost Transfer Update
- Question from the audience – Can you charge travel on a grant if not originally in the budget?
- Agenda:
- January 14, 2020 (there were no meetings in November or December, 2019) – Presentation Slides
- Agenda:
- May 2020 SPA Location Change
- NIH Public Access Policy
- IPAS Rebudget Requests
- NIH Multiple PI Certification
- NIH Human Subjects Decision Tool
- FDP Expanded Clearinghouse
- ORCiD
- NIH Submission Policies
- Agenda:
- October 15, 2019 – Presentation Slides
- Agenda
- NSF Audit
- New IPR Form
- IPR Highlight
- Effort on Federal Projects
- NIH Update
- SPS UConn Health Budget Templates
- Human Subjects Research
- New IPAS Form
- Change to Project Agreement Issuance
- Agenda
- September 17, 2019 – Presentation Slides
- Agenda
- Draft FDA Guidance with Dr. Wesley Byerly
- Fringe Benefits
- Use of Research Labs
- UConn Health OVPR Costs
- Training Updates
- Pre-Award Updates
- NIH Other Support
- NIH HFT Use
- ORCiD ID
- Sponsor Invoicing
- Cost Transfers
- Team Structure in SPS
- SPS Policy Updates
- Agenda
- May 28. 2019 – Presentation Slides and Compliance Slides
- Agenda
- CRA Study Group Update
- UCH Compliance Group (see compliance slides)
- Year End Processes
- 7630 Account Category
- Fringe Benefit Rates
- Salary Cap Updates
- Agenda
- April 30, 2019 – Presentation Slides
- Agenda
- Year End Dates
- Redesigned eRAWebsite
- NIH Funding Strategies
- Fringe Benefit Rates
- Salary Cap Updates
- Agenda
- March 26, 2019 – Presentation Slides
- Agenda
- Grant Writer Kit Bonin
- Training Update
- OIG Audit Overview
- NIH Updates
- Raises in July
- Agenda
- January 15, 2019 – Presentation Slides
- Agenda
- Training Update
- Export Control
- Fly America Act
- NIH Updates
- Agenda
- November 27, 2018 – Presentation Slides
- NIH Handouts
- Agenda
- New Website
- Training Update
- Review of FDP Agreement Request Form
- AMS Updates
- Transfer Vouchers
- Decoding NIH Grant Numbers (see NIH handouts)
- Sponsor Updates: NIH and NSF
- Agenda
- October 23, 2018 – Presentation Slides
- Agenda
- Training Update
- New Website
- Review of new ICF
- Procurement and Sole Sourcing
- Process for Contracts and Subawards
- Effort Reporting
- New F&A Agreement
- Sponsor Updates: NSF, NIH, and DOD
- Agenda
- May 2018
- March 12, 2018
- February 6, 2018
- November 8, 2017
- September 21, 2017
- May 24, 2017
- April 19, 2017
- March 8, 2017
- February 8, 2017
- January 11, 2017
- November 22, 2016
- October 19, 2016
- September 21, 2016
- June 15, 2016
- April 20, 2016
- March 2, 2016
- January 20, 2016
- November 18, 2015
- October 21, 2015
ResearchMatch
 ResearchMatch.org is a national online recruitment tool for health research, funded by the National Institutes of Health and maintained at Vanderbilt University. ResearchMatch connects health researchers with individuals interested in volunteering, through its secure online matching tool. UConn Health is part of the ResearchMatch Network. There is no cost to UConn Health researchers to use ResearchMatch. There are currently over 120,000 registered volunteers across the country. ResearchMatch is also now available in Spanish and simplified Chinese.
ResearchMatch.org is a national online recruitment tool for health research, funded by the National Institutes of Health and maintained at Vanderbilt University. ResearchMatch connects health researchers with individuals interested in volunteering, through its secure online matching tool. UConn Health is part of the ResearchMatch Network. There is no cost to UConn Health researchers to use ResearchMatch. There are currently over 120,000 registered volunteers across the country. ResearchMatch is also now available in Spanish and simplified Chinese.
To see if ResearchMatch might be a useful recruitment tool for your health-outcomes research study, use this link to register: https://www.researchmatch.org/researchers/. You do not need to be a Principal Investigator to register. The registration process takes less than 10 minutes. Once registered, you will be granted “feasibility access” to search volunteer aggregate data, including geographic and health demographics. Please note that you may not use ResearchMatch to recruit for a registry.
If you decide to use ResearchMatch to conduct participant recruitment, you will need IRB approval:
- The Vanderbilt IRB provides oversight for ResearchMatch as a recruitment tool and this has been documented within the ResearchMatch IRB Letter of Understanding (available upon request), but to use ResearchMatch as a recruitment tool for a specific study, you will first need to get UConn Health IRB approval.
- For the IRB submission, the following language may be used to describe ResearchMatch as a recruitment method: Potential volunteers will be contacted by ResearchMatch via an email contact message containing IRB-approved recruitment language for this study (not including direct study contact information such as email or phone number). Volunteers will then have the option of replying by clicking ‘yes’ or ‘no’ in the contact message. If a volunteer chooses to respond in the affirmative, they will authorize ResearchMatch to release their contact information to the PI (or ResearchMatch designee) who will be responsible for managing that information according to institutional guideline
- The contact message consists of the language to be used in the email sent by ResearchMatch on your behalf to potential participants. Please note that your contact message must not include your direct study contact information (email or phone), and must not exceed 2000 characters. If the study involves in-person participation, you may want to include the geographical location of the study site. If you intend to include healthy controls, specify that in your contact message, otherwise, a healthy control volunteer may decline participation. Please see contact message examples provided by ResearchMatch as well as this form that offers additional tips for creating an effective contact message.
- Once ResearchMatch is an IRB-approved recruitment method, you may then register as a researcher to request “recruitment access” in ResearchMatch. You will need to upload your IRB approval letter and your IRB-approved contact message. Recruitment access will give you the ability to send your contact message to potential participants that you select. If a potential participant agrees, you will then have access to his/her contact information in order to contact about possible study participation.
For questions about how to use ResearchMatch for your health research, please contact UConn/UConn Health ResearchMatch Liaison, Ellen Ciesielski, eciesielski@uchc.edu.
How to Register

2. On the PRS log-in page, in the Organization field, enter “UConnHealth” (no spaces) if your account was created under UConn Health or “UConn” if your account was created under UConn.
3. Click on the blue “Create New Record” button on the far right of the page. The system will walk you through the creation of the record. Data entered on previous pages will be retained so that you can return to complete the record at a later date.
The person who creates the record is the “Record Owner.” The Record Owner may be the Principal Investigator (PI) or someone designated by the PI to create/update the record on his/her behalf. The PI must perform the final review and approval of the record and each subsequent update in the PRS. The PI is responsible for ensuring that accurate information about the trial is entered and that updates to the record are completed in a timely manner (see table below for specific deadlines).
Complete the below fields as follows:
-
-
- Organization’s Unique Protocol ID: Enter the IRB number.
- Secondary IDs: Enter the grant number.
- Record Verification Date: Enter the month and year in which you are completing the record. This field will need to be updated each time you update the record after it is registered.
- Primary Completion Date: Enter the anticipated final data collection date, specifically regarding the Primary Outcome Measure.
- Responsible Party: Select the Principal Investigator from the drop-down menu.
- Collaborators: Add any other organizations providing support, including funding, design, implementation, data analysis and reporting.
-
For studies reviewed by UConn Health:
-
-
- Board Name: UConn Health IRB
- Board Affiliation: UConn Health
- Board Contact phone and e-mail: 860.679.8729; irb@uchc.edu
- Board Contact Address: UConn Health IRB, 263 Farmington Avenue, Farmington, CT 06030-1511
-
For studies reviewed by UConn Storrs and Regional Campuses:
-
-
- Board Name: UConn IRB
- Board Affiliation: UConn
- Board Contact phone and e-mail: 860.486.0986; irb@uconn.edu
- Board Contact Address: UConn IRB, 438 Whitney Road Extension, Unit 1246, Storrs, CT 06269
-
The Final Rule dictates that ClinicalTrials.gov record fields are updated on the below schedule:
| Data Field | Deadline for Updating (i.e., not later than the specified date) |
|---|---|
| Study Start Date | 30 calendar days after the first subject is enrolled (if the first human subject was not enrolled at the time of registration). |
| Intervention Name(s) | 30 calendar days after a nonproprietary name is established. |
| Availability of Expanded Access | 30 calendar days after expanded access becomes available (if available after registration); and 30 calendar days after an NCT number is assigned to a newly created expanded access record. |
| Expanded Access Status | 30 calendar days after a change in the availability of expanded access. |
| Expanded Access Type | 30 calendar days after a change in the type(s) of available expanded access. |
| Overall Recruitment Status | 30 calendar days after a change in overall recruitment status. If Overall Recruitment Status is changed to “suspended,” “terminated,” or “withdrawn,” the Why Study Stopped data element must be submitted at the time the update is made. |
| Individual Site Status | 30 calendar days after a change in status of any individual site. |
| Human Subjects Protection Review Board Status | 30 calendar days after a change in status. |
| Primary Completion Date | 30 calendar days after the clinical trial reaches its actual primary completion date. |
| Enrollment | At the time the primary completion date is changed to “actual,” the actual number of participants enrolled must be submitted. |
| Study Completion Date | 30 calendar days after the clinical trial reaches its actual study completion date. |
| Responsible Party, by Official Title | 30 calendar days after a change in the responsible party or the official title of the responsible party. |
| Responsible Party Contact Information | 30 calendar days after a change in the responsible party or the contact information for the responsible party. |
| Device Product Not Approved or Cleared by U.S. FDA | 15 calendar days after a change in approval or clearance status has occurred. |
| Device Product Not Approved or Cleared by U.S. FDA | 15 calendar days after a change in approval or clearance status has occurred. |
| Record Verification Date | Any time the responsible party reviews the complete set of submitted clinical trial information for accuracy and not less than every 12 months, even if no other updated information is submitted at that time. |
ClinicalTrials.gov
Over the past two decades, government agencies and the International Committee of Medical Journal Editors (ICMJE) have issued laws and directives on the subject of clinical trial registration. All parties have consistently agreed that the purpose of trial registration is to promote the public good by ensuring that the existence and design of clinical trials are publicly available. The registration effort began with the development of a publicly available website, ClinicalTrials.gov, a service of the National Institutes of Health (NIH), developed by the National Library of Medicine.
In 1997, the Food and Drug Administration (FDA)/NIH began requiring registration for only a limited number of trials. Then in September 2007, the FDA Amendments Act (Title VIII. Sec. 801) significantly expanded the scope of clinical trials that must be registered and set civil monetary penalties for failing to register “applicable trials.” In 2004, the ICMJE defined trials that must be registered in order to be considered for publication in journals that adhere to ICMJE standards. In 2007, the ICMJE expanded the definition of trials that must be registered. Scores of journals (not limited to medical journals) have adopted the registration policy. In 2016, the final rule for Clinical Trials Registration and Results Information Submission (42 CFR Part 11), which clarifies and expands the requirements in FDAAA 801, was released, and the NIH Policy on the Dissemination of NIH-Funded Clinical Trial Information was published.
The bottom line:
The FDA, NIH and the ICMJE each have their own requirements for registration, and while some of the requirements overlap, there are some differences. In order to comply with the law and preserve the ability to publish in many journals, all sets of requirements must be met. UConn Health is committed to supporting Principal Investigators (PIs) with meeting the requirements concerning the public availability of clinical trial data in ClinicalTrials.gov, as outlined in our University ClinicalTrials.gov policy.
How is “clinical trial” defined and what are the registration deadlines?
According to NIH:
A clinical trial is a research study1 in which one or more human subjects2 are prospectively assigned3 to one or more interventions4 (which may include placebo or other control) to evaluate the effects of those interventions on health-related biomedical or behavioral outcomes.5 All clinical trials must be registered within 21 days of enrollment of the first participant.
1See Common Rule definition of research at 45 CFR 46.102(d).
2See Common Rule definition of human subject at 45 CFR 46.102(f).
3The term “prospectively assigned” refers to a pre-defined process (e.g., randomization) specified in an approved protocol that stipulates the assignment of research subjects (individually or in clusters) to one or more arms (e.g., intervention, placebo, or other control) of a clinical trial.
4An intervention is defined as a manipulation of the subject or subject’s environment for the purpose of modifying one or more health-related biomedical or behavioral processes and/or endpoints. Examples include: drugs/small molecules/compounds; biologics; devices; procedures (e.g., surgical techniques); delivery systems (e.g., telemedicine, face-to-face interviews); strategies to change health-related behavior (e.g., diet, cognitive therapy, exercise, development of new habits); treatment strategies; prevention strategies; and, diagnostic strategies.
5Health-related biomedical or behavioral outcome is defined as the pre-specified goal(s) or condition(s) that reflect the effect of one or more interventions on human subjects’ biomedical or behavioral status or quality of life. Examples include: positive or negative changes to physiological or biological parameters (e.g., improvement of lung capacity, gene expression); positive or negative changes to psychological or neurodevelopmental parameters (e.g., mood management intervention for smokers; reading comprehension and /or information retention); positive or negative changes to disease processes; positive or negative changes to health-related behaviors; and, positive or negative changes to quality of life.
Registration Deadline: The NIH Policy on Dissemination of NIH-funded Clinical Trial Information applies to applications for funding submitted to NIH on or after 1/18/17. Registration is required no later than 21 days after the first patient is enrolled. The consent form for the trial is required to include the following language: “A description of this clinical trial will be available on http://www.ClinicalTrials.gov, as required by U.S. Law. This Website will not include information that can identify you. At most, the Website will include a summary of the results. You can search this Website at any time.”
According to the ICMJE:
A clinical trial is any research project that prospectively assigns people or a group of people to an intervention, with or without concurrent comparison or control groups, to study the cause-and-effect relationship between a health-related intervention and a health outcome. Health-related interventions are those used to modify a biomedical or health-related outcome; examples include drugs, surgical procedures, devices, behavioral treatments, educational programs, dietary interventions, quality improvement interventions, and process-of-care changes. Health outcomes are any biomedical or health-related measures obtained in patients or participants, including pharmacokinetic measures and adverse events. Those who are uncertain whether their trial meets the expanded ICMJE definition should err on the side of registration if they wish to seek publication in an ICMJE journal.
Registration Deadline: Registration at or before the time of first patient enrollment is as a condition of consideration for publication in an ICMJE journal.
According to the FDA:
The U.S. Department of Health and Human Services in September 2016 issued a final rule that specifies requirements for registering certain clinical trials and submitting summary results information to ClinicalTrials.gov. The new rule expands the legal requirements for submitting registration and results information for clinical trials involving U.S. Food and Drug Administration-regulated drug, biological, and device products. The FDA Amendments Act (Title VIII. Sec. 801) requires registration for all “applicable clinical trials,” regardless of funder, that are:
- Trials of drugs and biologics: Controlled, clinical investigations, other than Phase I investigations, of a product subject to FDA regulation, and
- Trials of devices: Prospective clinical studies of health outcomes comparing an intervention with a device against a control in human subjects (other than small clinical trials to determine the feasibility of a device, or clinical trials to test prototype devices where the primary outcome measure relates to feasibility and not to health outcomes); and pediatric postmarket surveillance studies, as required under the Federal Food, Drug and Cosmetic Act.
Registration Deadline: The FDA Amendments Act of 2007 requires that all trials, regardless of sponsor, must be registered in full no later than 21 days after the first patient was enrolled.
Who is responsible for registering a trial?
The PI is ultimately responsible for determining that registration requirements are met. Although some industry sponsors will do the actual registration work, it is still the PI’s responsibility to ensure that the registration has been accomplished and is accurate.
What are the penalties for failing to register?
According to the ICMJE:
Unregistered trials will not be considered for publication in journals that adhere to ICMJE standards. Questions about policies of a specific journal should be addressed to that journal directly.
According to the FDA/NIH:
Penalties may include civil monetary penalties up to $14,724 fine for failing to submit or for submitting fraudulent information to ClinicalTrials.gov. After notification of noncompliance, the fine may go up to $14,724 per day until resolved. For federally-funded grants, penalties may include the withholding or recovery of grant funds.
Who can help with questions about meeting the requirements?
To comply with University policy regarding ClinicalTrials.gov, please direct any questions to the local Protocol Registration and Results System (PRS) Administrator for UConn/UConn Health, Ellen Ciesielski, Research Integrity & Compliance. The local PRS Administrator serves as a point of contact for resolving requests by ClinicalTrials.gov and regularly monitors to promote compliance with the requirements.
IACUC Membership
1. PURPOSE
1.1 This policy establishes the responsibilities and roles of IACUC members, as well as the processes to add and remove an IACUC member to the roster.
2. IACUC MEMBERSHIP
2.1 IACUC members shall be selected in accordance with Public Health Service Policy on Humane Care and Use of Laboratory Animals Policy IV.A.3, IV.B., and IV.A.C , Animal Welfare Act Regulations 2.31, and the “IACUC Constitution and Function” detailed in The Guide for the Care and Use of Laboratory Animals. PHS policy is regulated by the Office of Laboratory Animal Welfare (OLAW) and the Animal Welfare Act is regulated by the United States Department of Agriculture Animal and Plant Health Inspection Service (USDA-APHIS). IACUC members will be selected based on qualifications, education, and/or experience and a positive attitude toward IACUC membership.
2.2 Membership is contingent upon commitment to items listed in section 2.4 of this policy and subject to review by the IACUC executive staff (Institutional Official [IO], IACUC Chair, IACUC Vice Chair, and the Director). Scientific members shall be appointed to a 3-year term subject to the above-mentioned annual performance review. Appointments can be renewed. IACUC members shall inform the IACUC Administrator by email that they accept their appointment when they receive the appointment letter from the IO.
2.3 Program Responsibilities of the IACUC as defined by the above regulations
2.3.1 Ensure compliance with all regulatory agencies and AAALACi
2.3.2 Semi-annual review of the program and inspection of all facilities and prepare reports of results to the Institutional Official
2.3.3 Review of all activities involving live animals
2.3.4 Review, and investigate if necessary, all concerns involving live animal care and use.
2.3.5 Making recommendations to the Institutional Official regarding any aspect of the animal care and use program
2.3.6 Suspending activities as required
2.3.7 Oversight of training programs related to animal care and use
2.3.8 Oversight of occupational health and safety protocols related to animal care and use
2.3.9 Oversight of policies pertaining to animal care and use
2.3.10 Meeting as necessary to ensure required activities occur
2.3.11 Advise on facility security as related to animal care and use
2.3.12 Advise on emergency and disaster planning as related to animal care and use
2.3.13 Conduct post-approval monitoring activities
2.4 Individual IACUC member responsibilities
2.4.1 Review all materials provided for IACUC meetings
2.4.2 Familiarize yourself with all protocols on he agenda for a full committee review (FCR)
2.4.3 Declare any potential conflict of interest related to their work on the IACUC; if there is a conflict of interest during review, recuse yourself from the vote
2.4.4 Be aware of federal regulations in the Animal Welfare Act, Title 7, Chapter 54, section 2157 – it is unlawful for any member of an IACUC to release confidential information to a third party or to use, or attempt to use, confidential information to their own advantage.
2.4.5 Complete all training and continuing education sessions as assigned by the IACUC Administrator
2.4.6 Attend a majority of the scheduled committee meetings
2.4.7 Demonstrate a professional and courteous manner towards other members, staff, and faculty.
2.4.8 Familiarize yourself with applicable policies, regulations, and guidelines given to you in your “IACUC Member Handbook”.
2.4.9 Act as a liaison for your colleagues
2.4.10 Complete all assigned protocol reviews by the due date assigned by the IACUC administrative staff. If you cannot, contact the IACUC Administrator so another reviewer can be assigned.
2.5 Individual IACUC member rights
2.5.1 Request FCR for any animal activity for any reason within the timeframe given to you.
2.5.2 Voice a minority opinion for the record related to any topic covered in the semi-annual report to the IO.
2.5.3 Participate in any scheduled facility inspection or program review activity.
2.5.4 Raise policy issues to the Committee for discussion
2.6 Expectations of Committee members at meetings
2.6.1 Share your unique input to get all the issues “on the table”. Members are expected to be active participants in IACUC discussions; for instance, if you have information that has not been discussed, share it.
2.6.2 Think critically but fairly and use the criteria for approval (Appendix I) to decide whether to approve the research. If you have a concern, problem, or recommended change, be able to base it on the criteria for approval.
2.6.3 Listen and learn from the group but think and vote independently. Vote according to your own conscience in an informed manner.
2.6.4 Respect minority opinions when offered.
2.6.5 Remember that all discussions and Committee decisions are considered confidential.
2.6.6 Understand that Committee decisions are made by majority rule and not consensus.
2.7 IACUC Chair
2.7.1 Responsibilities
2.7.1.1 Assigns/Approves reviewers for protocols meeting the criteria for a Designed Member Review
2.7.1.2 Signs OLAW Inter-Institutional Assurances
2.7.1.3 Runs the IACUC meetings
2.7.1.4 Has the authority to make exemptions to IACUC Policies
2.7.1.5 Assigns members to subcommittees when required
2.7.1.6 Any other as defined by laws, regulations, guidelines, or policies
2.7.2 The IACUC Chair should normally be an IACUC member who is a respected individual with knowledge of research ethics, regulations, and guidance that govern animal research.
2.7.3 The IACUC Chair will be appointed to a 3-year term.
2.8 IACUC Vice Chair
2.8.1 Responsibilities
2.8.1.1 Discharge the IACUC Chair’s responsibilities when the Chair is unable to do so
2.8.1.2 Discharge the responsibilities assigned by the IACUC Chair
2.8.1.3 Assist in the operation of the IACUC
2.8.2 Appointment of the Vice Chair is made by the IACUC Chair and the IACUC Administrator wit the approval of the IO.
2.8.3 The IACUC Vice Chair should commit to a 3-year appointment unless renewed or appointed to become IACUC Chair, thereupon the responsibilities and appointment term will be exclusive as defined above.
3. Process for Appointment of IACUC Members
3.1 Process of nomination of new members
3.1.1 The IACUC Chair, Vice Chair, Director, and/or Administrator identify and nominate an individual for a vacancy on the IACUC.
3.1.2 The IACUC Administrator obtains the resumé or curriculum vitae of the nominee and forwards to the IACUC Chair and IO for review
3.1.3 If all parties agree that the background of the potential member is a good fit with the current membership of the IACUC, an interview (telephone, web, or in-person) will be conducted with the IACUC Chair, Vice Chair, Director, Administrator, and the IO.
3.1.4 After the interview, a decision by all parties will be made regarding the appointment.
3.1.5 Once the nominee has been appointed, the IACUC Administrator prepares and appointment letter which is signed by the IO and send to the individual.
3.1.6 Once the nominee has accepted the appointment, a meeting will be held between the new member and the IACUC Administrator who assigns any training and provides the individual with an “IACUC Member Handbook”. Training, documents given, etc. are all recorded on a new member orientation form held by the IACUC office. The individual is added to the IACUC roster.
3.1.7 The new IACUC member is assigned a mentor – typically, a seasoned IACUC member who will help guide the new member with regard to protocol reviews and provide any other help the new member may need.
3.2 Process for reappointment of members
3.2.1 Members are appointed to a three-year term, subject to annual continuation. After the annual evaluation of the member (see section 2.2 above), if all parties agree the member should continue for the next year, a continuation appointment letter will be prepared by the IACUC Administrator and sent to the IACUC member.
4. Process for Removal of IACUC Members
4.1 The IO or their designee is responsible for deciding whether to remove an IACUC member.
4.2 If the IACUC member will no longer serve as an IACUC member, the IACUC administrator will prepare a thank you letter which will be signed by the IO and sent to the member.
4.3 The membership roster will be updated as required.
Effective Dates: December 5, 2024 through December 31, 2027
Appendix I:
Criteria for Reviewing Animal Care and Use Protocols
Compiled from regulations set forth from:
* Animal Welfare Act (Title 7, Chapter 54, United States Code)
* Animal Welfare Regulations (9 CFR, Chapter 1, subchapter A, parts 1-4)
* PHS Policy on the Humane Care and Use of Laboratory Animals
* US Government Principles
I: Selection and Justification of Species
- The animal model selected should be the most appropriate species for the project based upon anatomical, physiological, or other characteristics in consideration of the scientific objectives and the need to obtain valid results
II: Living Conditions of the Animals
- The living conditions of the animals must be appropriate for their species and contribute to their health and comfort
- Any deviations from standards set forth by the Guide and the USDA regulations must be scientifically justified and approved by the ACC
III: Justification for the Use of Animals
- Procedures involving animals should be designed and performed with due consideration given to their relevance to human or animal health, the advancement of knowledge, or the good of society
- Protocols involving animals should have sound research design and should yield valid results
- Projects must not unnecessarily duplicate previous experiments and any duplications must be justified
IV: Application of the Principles of Replacement, Reduction, and Refinement
- Replacement: when objectives can be achieved using reasonably available non-animal models, the alternative should be used
- Reduction: the number of animals used should be the minimum necessary to achieve scientific goals
- Refinement: procedures should be used that have the least amount of potential pain, discomfort, distress, or morbidity
-PI must provide a written narrative describing the methods and sources used to determine that alternatives to painful/distressful procedures were not available:
Database search* (must be >1) if utilizing USDA-regulated species must include:
Databases searched
Date of search
Years covered by search
Search strategy used
*Other sources consulted (e.g., experts in field, standard reference books, etc.)
V: Ethical Cost-Benefit Relationship
- The ethical cost of the research must be outweighed or balanced by the potential value of research to human or animal health, the advancement of knowledge, or the good of society
VI: Treatment of Pain and Discomfort
- Unless the contrary is established, investigators should consider that procedures that cause pain or distress in human beings may cause pain or distress in animals
- Procedures that cause more than momentary pain or distress must be performed with state of the art sedation, analgesia, or anesthesia unless withholding is justified for scientific reasons
- Pain alleviating agents should be administered as part of a continuum when appropriate
VII: Post-Procedure Monitoring
- All animals must be monitored at appropriate intervals dictated by the nature of procedure, degree of potential pain, possible complications of the procedure
- During monitoring, animals should be evaluated for the presence of pain, discomfort, or distress
- Observer should use criteria based upon the normal behavior pattern of the species
- Simple observation by a skilled observer can reveal a great deal of information
VIII: Restraint
- Mechanical restraint must be justified
- Unacceptable to use mechanical restraint on awake animals if non-mechanical forms can be used
- Device must provide the animal with the opportunity to assume its normal postural adjustments
- Animals should be conditioned to the device
- Duration of restraint must be minimized
- Animal should be observed at appropriate intervals
IX: Euthanasia
- Method of euthanasia should be based upon the species, the size of the animal, the scientific objectives of the experiment, and its ability to quickly and painlessly produce a loss of consciousness and death
- Euthanasia must comply with current (2000) American Veterinary Medical Association Panel on Euthanasia Guidelines
X: Qualification of Research Personnel
- Personnel who perform procedures involving live animals must be
– Knowledgeable about the biology of the species being used
– Fully qualified by training and experience to carry out their assigned duties - Any individual who lacks the prerequisite qualifications must be trained before they interact with animals
XI: Occupational Health and Safety
- The ACC must be satisfied that there is an effective occupational health and safety program for all individuals using laboratory animals
– Ensures risks associated with experimental use of animals are reduced to acceptable levels
– Ensures personnel are appropriately trained with the use of any hazardous agents